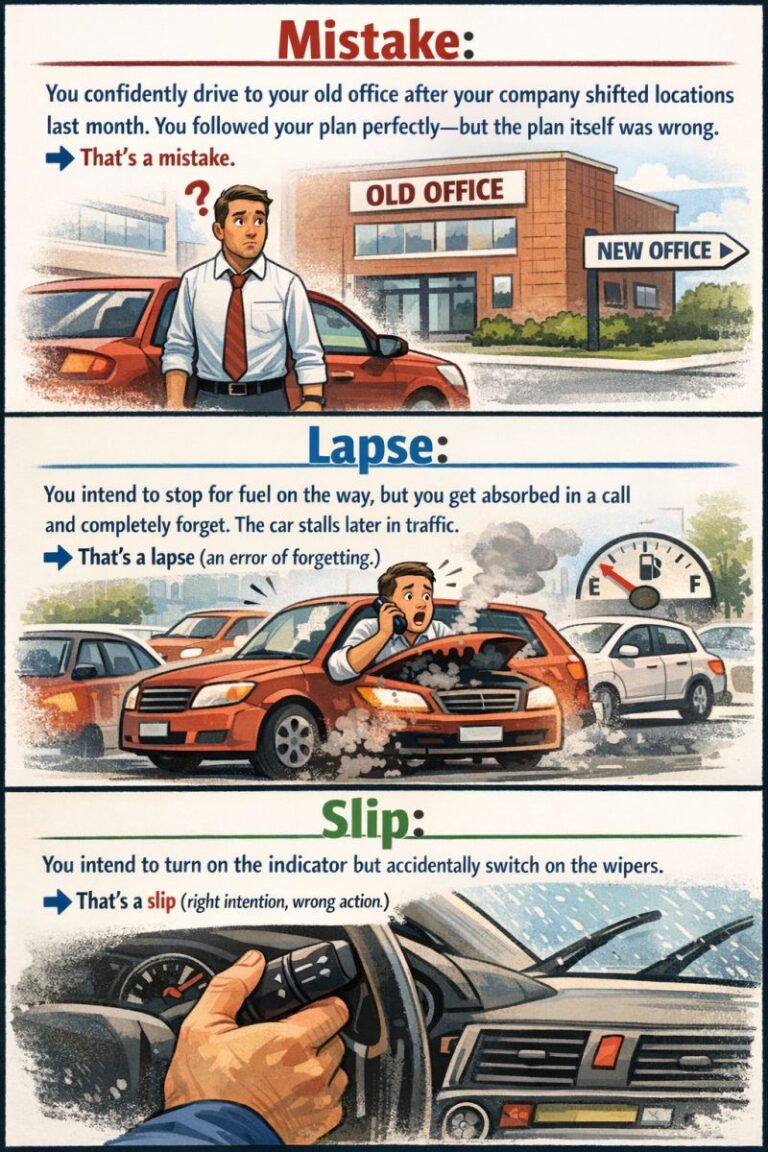
Minimize Human Error.
Maximize Profitability.

What You Need To Know About Our Company
At Blue Flame Consulting, we are dedicated to advancing the standards of pharmaceutical manufacturing through proactive error risk reduction strategies. Our team, composed of industry experts, leverages deep insights and cutting-edge methodologies to enhance operational safety and efficiency.
We specialize in identifying and mitigating human error in manufacturing, quality control, warehouse operations, ensuring our clients achieve the highest levels of reliability and excellence.
How we solve the problem using Process Streamlining
Our commitment is to transform the pharmaceutical industry, setting new benchmarks in safety and operational practices.
Clarify Process
Making activities and their context explicit
Risk Review
Identification and assessment of risk of errors
Understand Causes
Identification of risk influencing factors
Toolbox Error Reduction
Elimination or reduction of risk influencing factors
Continuous Reliability improvement
Continuous improvement in human reliability
Business Strategy
Evaluation on how companies can reduce costs and bring efficiency in their operations
Directly from the source
The FDA’s observation highlights the difference between less robust and more robust quality systems in pharmaceutical manufacturing. Less robust systems primarily focus on retraining personnel as a solution to human error, essentially repeating the same training that employees have already received.
How the process works
The phases are structured as:
Planning
Discovery
Ideate
Define
Validate
spanning over a timeline of approximately 12 weeks.
Identification of Pain Areas: Recognizing critical issues in processes.
Data Analysis & ERR Process: Analyzing data to understand and refine ERR strategies.
Problem Assessment & Prioritization: Evaluating and prioritizing issues for effective resolution.
Problem Assessment & Prioritization: Evaluating and prioritizing issues for effective resolution.
Opportunities and Solutioning: Identifying and implementing potential solutions.
Client Experience
Leading Indian pharmaceutical manufacturer (formulations and API operations)
Major export-focused pharmaceutical company serving regulated markets
Client identities are not disclosed due to confidentiality commitments.
Insights and News
Latest thinking on Error Risk Reduction (ERR) and Human Reliability in Pharmaceutical Manufacturing.
More than 27 years of experience including senior leadership role in pharmaceutical industry in the field of validation, training, corporate quality audit and quality management systems.
- Contributed to the field of human error investigation that resulted in significant reduction of human errors across sites.
- Formulated multiple corporate guidelines on QMS along with one on human error reduction.
- Authored IPA (Indian Pharmaceutical Alliance) guideline on Human Error Reduction.
- Spokesperson for global initiatives such as best practices sharing within the organisation and on various public forums.
- Successfully faced multiple regulatory inspections over the years.
- Passionate about fostering a culture of safety and operational excellence in the pharmaceutical industry through a focus on human error reduction.




What our Customer has to say about us....
Rajiv Kumar (Bhardwaj)
AVP and Site Quality Head, Ajanta Pharma
The team brought a structured and practical approach to reviewing our QC laboratory processes, with a strong focus on GEMBA and workflow simplification. The initiatives have been well received by analysts and have improved clarity and ease of execution in day-to-day operations. Overall, it has been a collaborative and value-adding engagement with a positive impact on process understanding.
A major strength of their approach is the focus on doer-level engagement ensuring that recommendations are not just theoretical but implementable.
Frequently Asked Questions (FAQs)
How can pharmaceutical companies promote a culture of compliance and error prevention?
Pharmaceutical companies can promote a culture of compliance and error prevention by providing ongoing training, conducting regular audits, implementing robust documentation systems, encouraging open communication about errors, and ensuring that employees understand the importance of their work in patient safety.
What measures are taken to prevent medication mix-ups and packaging errors?
To prevent medication mix-ups and packaging errors, pharmaceutical companies use barcoding systems, automated packaging equipment, color-coded labels, and visual inspection systems. Verification processes are also in place to confirm that the correct medication and dosage are being packaged.
What is the role of automation in reducing human errors in pharmaceutical manufacturing?
Automation plays a significant role in reducing human errors by automating repetitive and critical tasks, such as drug formulation and packaging. Automated systems can minimize dosage discrepancies, contamination risks, and labeling errors.